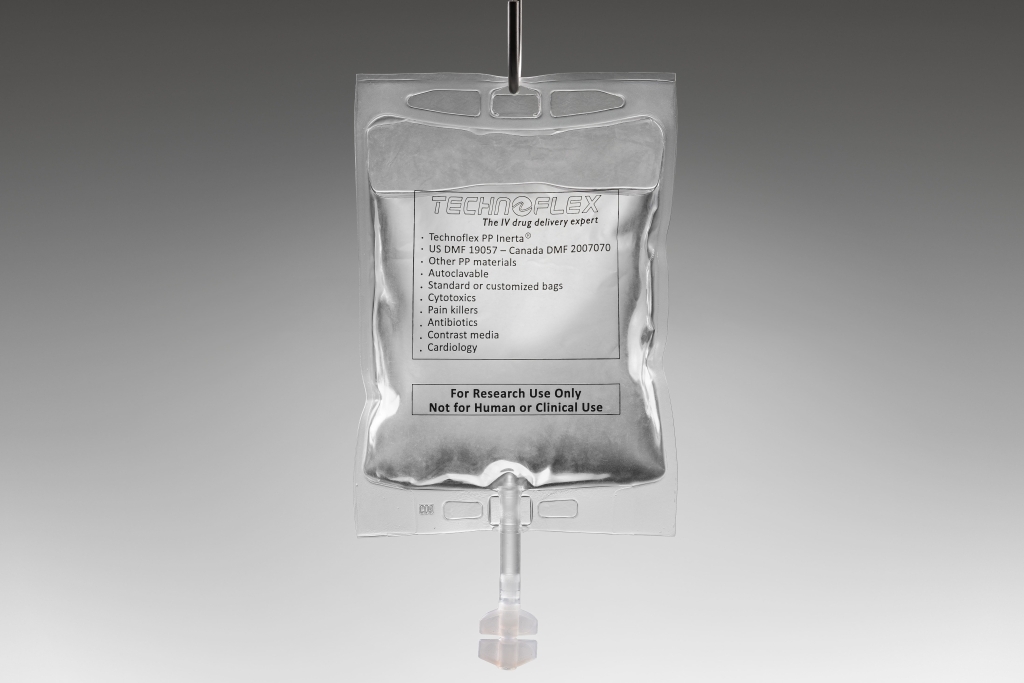
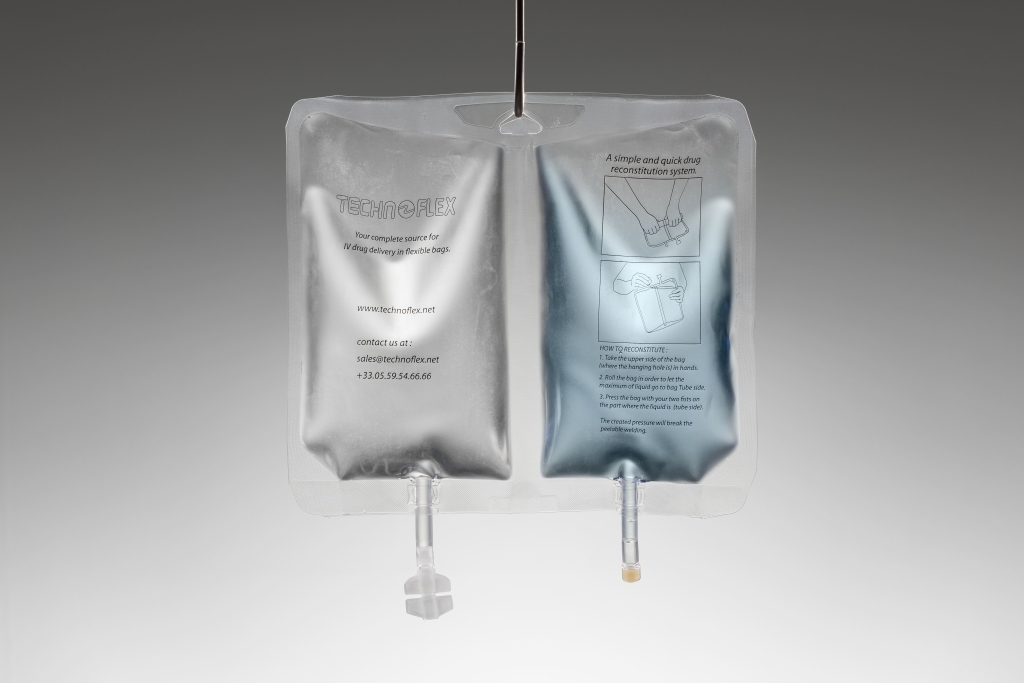
Technoflex, your flexible medical packaging expert
At Technoflex, we partner with the pharmaceutical and biotech industries to bring flexible medical solutions to life. From concept to final product, we design bags, components, and administration systems that are safe, reliable, and ready to use.
Through advanced technologies and continuous development, we anticipate tomorrow’s medical needs while staying focused on what matters most: the safety and care of patients and healthcare professionals.
For over 40 years, we have transformed expertise into meaningful solutions, combining performance, responsibility, and creativity to support the future of medical and biotech care.
Our history
From its early beginnings, Technoflex has grown into a trusted industrial partner for the pharmaceutical and biotech industries. Our journey has been shaped by strong technical expertise, high quality standards, and a constant drive to improve and evolve.
Our values
Commitment
At Technoflex, commitment means taking responsibility for projects from start to finish. We work closely with our customers, stay available during each phase, and follow through on technical and operational details. Our objective is simple: deliver what is agreed, under the expected conditions.
Reactivity
We organize our teams and processes to answer and act quickly. Requests, changes, and technical issues are handled directly by qualified contacts, allowing short decision paths and practical responses.
Expertise
With more than 40 years of experience, Technoflex combines proven technical expertise with a fully integrated in-house organization: a 15+ person R&D team, an internal laboratory, specialists in methods and industrialization, and a customer-focused commercial team. Together, these capabilities allow us to base our technical decisions on solid data, real production conditions, and direct customer feedback.
Quality is at the heart of our injectable solutions. We manufacture pharmaceutical packaging and components in compliance with GMP requirements and international regulations, ensuring safety, integrity and product purity.
Innovation is at the core of Technoflex’s development. Our R&D team designs advanced solutions for pharmaceutical and biotechnology applications, combining technical expertise with a deep understanding of customer needs.
At Technoflex, our customers are at the heart of every decision we make. Our Sales, Customer Service, and Supply Chain teams work together to provide a fluid, responsive, and human‑centered experience.
Technoflex supports pharmaceutical and biotechnology companies in meeting international regulatory requirements.
Through ISO 15378 and ISO 9001 certified quality systems, we continuously improve our processes to meet the highest pharmaceutical standards.
ISO 13485 – Medical devices
MDSAP US and CANADA
ISO 15378-2017 with GMP requirements applied to Primary packaging for drugs

We specialize in tailor-made solutions, developing custom-designed bags and connectors precisely adapted to each application. Working closely with our customers, we create bespoke products that meet specific technical and regulatory requirements, from early development to industrial production.
Our innovation approach also integrates eco-design principles, optimizing materials and processes to reduce environmental impact while maintaining the highest quality and performance standards.

From on‑site visits that bring our sales representatives directly into your environment, to a customer service team that listens, adapts, and responds quickly, and a supply chain designed for reliability and agility, we go beyond expectations to ensure every customer feels supported, understood, and genuinely cared for.

Our dedicated in-house regulatory affairs team provides personalized support for regulatory submissions, including Marketing Authorization Applications (MAA). We supply the technical and regulatory documentation required to support your approval process.
Through expert guidance and close collaboration, we help ensure efficient submissions and full regulatory compliance.