News & Events
Follow our latest news, industry highlights, and event announcements. Stay informed about what’s happening within our company and across the pharma and biotech sectors.
-
 News
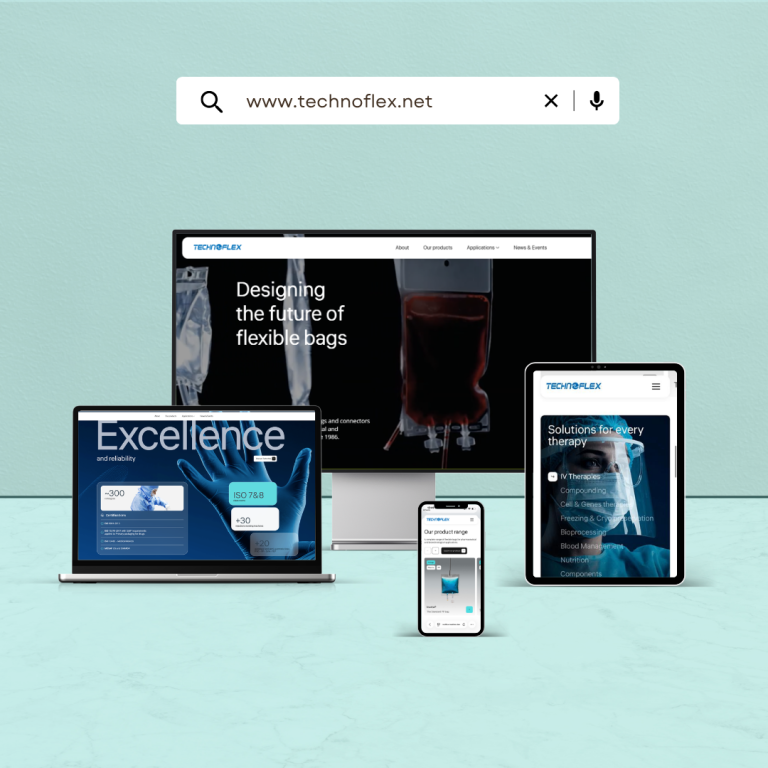
NewsOur website gets a fresh new look!
Read the article -
 News
NewsOur CSR Approach on Dinamika
Read the article -
 Events
EventsCPHI CHINA
Read the article -
News
EcoVadis Silver Label
Read the article -
 Events
EventsASHP MIDYEAR
Read the article -
 Events
EventsHEALTHCARE PACKAGING EXPO
Read the article -
 Events
EventsAABB
Read the article -
 Events
EventsCPHI WORLDWIDE
Read the article -
 Events
EventsADVANCED THERAPIES WEEK EUROPE
Read the article -
 Events
EventsCPHI INDIA
Read the article
Have a question or a project in mind?
Our team is here to help. Fill in the form and we’ll get back to you shortly.

